Shining a light on molecular switches
First structural insights obtained from femtosecond X-ray crystallography
Fluorescent proteins, in particular those that can be turned on and off, are indispensable tools in modern cell biology. Researchers at the Max Planck Institute for medical Research in Heidelberg, together with colleagues from several groups in France, have now succeeded in observing the steps in this switching process and in using the results to create a modified protein with improved properties for biological applications.
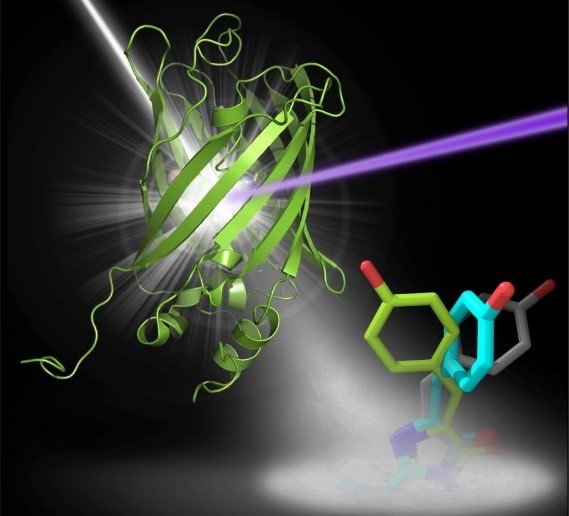
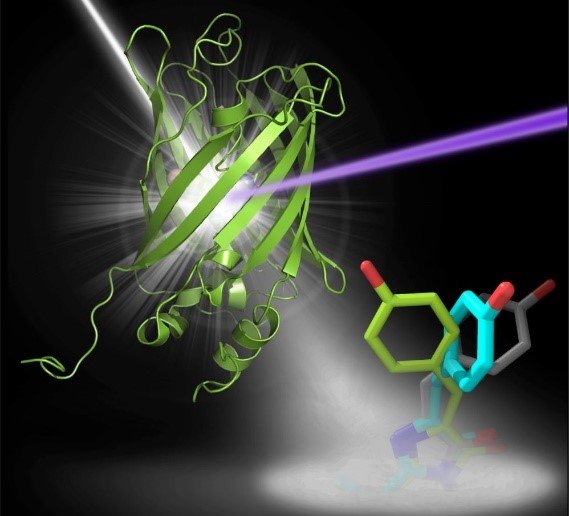
Fluorescent molecules, when exposed to light of a particular wavelength, emit light of a different wavelength. This property makes them indispensable in the study of cellular processes by light microscopy, and they have led to dramatic progress in understanding the cell in health and disease. The fluorescence stems from a specific area of the molecule, the chromophore. Particularly valuable are fluorescent proteins whose fluorescence can be turned on and off rapidly and specifically as required. These must therefore exist in two different conformations, only one of which is fluorescent, which can be interconverted by a trigger from outside. For the study of rapid cellular events, the switching must also be very fast, and fluorescence effects generally meet this requirement, occurring on a femto- to picosecond time-scale. An important example is the protein rsEGFP2, one of a large family of synthetic variants of GFP, a naturally occurring fluorescent protein of certain jellyfish.
The researchers in Heidelberg, led by Ilme Schlichting, and their cooperation partners have studied the process by which rsEGFP2 shifts between its fluorescent and non-fluorescent states (in this case by irradiation with light of specific wavelengths). This has revealed the structures of the intermediates via which the conformation changes between on and off.
Designing improved molecules
This knowledge is vital when it comes to designing new molecules for ever more applications. In the present case, the new knowledge of the details of the switching pathway suggested roles for individual amino-acids in the vicinity of the chromophore of rsEGFP2, and that replacing one of these with a larger chain might increase the efficiency of the switching process, which would be a desirable property for practical use of rsEGFP2. This prediction proved to be correct: the transition between the on and off states was substantially facilitated.
Observing the details of the exceedingly rapid structural changes in the chromophore region was possible only through the use of femtosecond X-ray crystallography. This technique has been developed in the last 7 years, with crucial contributions from the Heidelberg group. The measurements were made using the SLAC free-electron laser in Stanford (USA). In this method, thousands of microcrystals of the sample are passed through a beam of pulsed X-rays, whose pulses are of extreme intensity yet extremely short (femtosecond) duration. The patterns of diffracted X-rays that are recorded can then be ascribed to molecules in different stages of a correspondingly fast (femtosecond) transition and used to determine the respective structures of the chromophore. The success of the present work towards the design of improved fluorescent probes is likewise a further triumph for this revolutionary technique of structural biology.